An often mentioned simple way to improve water electrolysis is to modulate the current, for example, by rapidly switching it on and off periodically. Many papers report improvements without a fair comparison with the non-modulated (DC) case. In such a comparison, the same amount of hydrogen is produced per unit time, so that the average current over time is equal.
Unfortunately, doing exactly this, we found no positive effect of modulating the current in the 0.01-1000 kHz range.

This is easy to understand, because energy losses usually increase more than linearly with current density: to obtain the same average current density, the lower energy losses at lower-than-average current are more than offset by the higher energy losses during periods of higher-than-average current.
There are a few noteworthy exceptions: for example, at low currents, the stack losses can decrease with increasing current density due to shunt currents. Also, by modulating at the time scale of gas build-up, a small advantage may be obtained. Finally, there may be a regime at extremely high frequencies at which the catalysis will be influenced. However, in general, claims of improvement should be considered with healthy scepticism.
For a cell voltage V0+ARj, the larger the difference between the high current (jhigh) and low current (jhigh), and the larger the fraction of the time D or 1-D that the current is high or low, the higher the specific energy consumption (SEC) compared to DC (jav) :
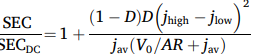
This prediction was nicely validated by in-house experiments with an AEM cell.



